All About Polyester (PET) Plastic
Polyester, in most cases, refers to the trade name for polyethylene terephthalate or PET. It is the most used thermoplastic polymer in the world. In fact, it makes up about 18% of the world’s polymer production. The textile industry uses the term polyester a lot. It is used to make clothing fibers and packaging of the textile products. The other popular use of Polyester is in making plastic PET bottles. Polyester is a transparent semi-crystalline material which also happens to have a high barrier to moisture. It also has a favorable strength-to-weight ratio.
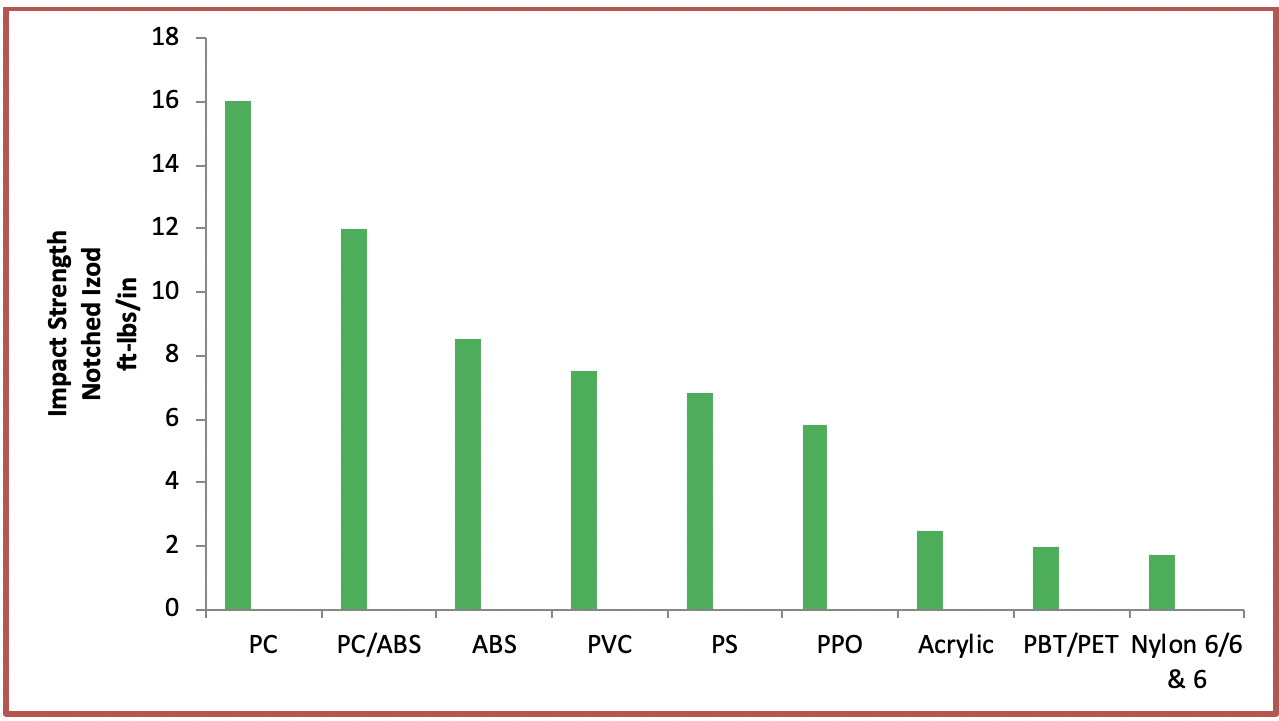
Polyester or PET has good chemical resistance and does not react with foods and liquids. It is also a shatter-proof material and hence can replace glass when used to make containers. Being a thermoplastic material, it has a melting point of 260oC. It can be heated to that temperature, cooled, and then reheated again without any significant degradation. The ability of polycarbonate to liquefy via reheating makes it a good fit for injection molding. On the commercial front, it is widely available and reasonably priced.
History of Polyester
The earliest record of polyester creation dates back to 1940 when DuPont wanted to make textile fibers using polyester. Meanwhile, in England, PET was patented by a couple of employees of the English company Calico Printers Association Ltd. DuPont trademarked its creation with the name Mylar and received registration in the year 1952.
Meanwhile, in the Soviet Union, PET was manufactured at the Institute of High-Molecular Compounds in the year 1949. This institute was part of the USSR Academy of Sciences. And finally, the PET bottle was patented by Nathaniel Wyeth in 1973. So, overall, polyester and PET has had a very global evolution.
Today, the global production for polyester is almost 80 million tons per annum.
Manufacturing of Polyester
Polyester can be produced via two processes, one is using ethylene glycol and dimethyl terephthalate, and the second is using ethylene glycol and terephthalic acid.
In the first scenario, dimethyl terephthalate and ethylene glycol are allowed to react in the presence of a basic catalyst at a temperature of about 150oc to 200oc. Methanol is distilled out and then excess ethylene glycol is distilled with the help of vacuum. This process of producing polyester is also known as the transesterification process. It involves two steps and the chemical reactions of those two steps are as shown below:
First step
C6H4(CO2CH3)2 + 2 HOCH2CH2OH → C6H4(CO2CH2CH2OH)2 + 2 CH3OH
Second step
n C6H4(CO2CH2CH2OH)2 → [(CO)C6H4(CO2CH2CH2O)]n + n HOCH2CH2OH
In the second scenario, ethylene glycol and terephthalic acid are allowed to react at high temperature and moderate pressure in a process known as esterification. Water is removed via distillation during the process. The chemical reaction for this process is as follows:
n C6H4(CO2H)2 + n HOCH2CH2OH → [(CO)C6H4(CO2CH2CH2O)]n + 2n H2O
Sometimes, polyester or PET suffers from degradation in the above processes. If that happens, then certain copolymers or stabilizers are used.
Advantages of Polyester
Polyester is available in different grades depending upon the application where it is to be used. It is available as a homopolymer and a version created by modification with copolymers. When used in textiles and for making fabrics, it is known as polyester. When used in packaging, it is known as PET. It was also known as PET-P at a point in time. Whatever its name, polyester has many advantages such as:
- Polyester is transparent and has good transmissivity
- Polyester is low cost and readily available
- Polyester is resistant to moisture and has low water absorption
- Polyester has great electrical properties
- Polyester has good chemical resistance to water as well as organic material
- Polyester can be recycled and broken down into a resin form
- Polyester has good heat and aging resistance
Applications of Polyester
Clothing: Polyester is quite well-known for making garments and clothing items. It has a good moisture barrier and is a great material for making sports and weather-resistant clothing. It is also used in combination with cotton to produce wrinkle-free clothing. It is more resistant to tears than cotton or other materials that are also used to make clothes.
Home Improvement: Polyester is used extensively in making furniture and upholstery. It is also used to make carpets, as it is available in a fiber form. Ropes used in domestic applications are often made using polyester.
Packaging: Polyester and PET are both used extensively in packaging applications. The most ubiquitous of those applications is the PET bottle. It is used to pack a wide variety of beverages, from soft drinks to juices. Food products are also packed using PET jars. Certain types of trays (egg boxes) are made using polyester. Certain types of packaging films are also made using polyester.
Tapes: PET is used as a carrier for magnetic tapes and also in pressure-sensitive adhesive tapes because of its high mechanical strength.
Food: Crystallizable PET can withstand high oven temperatures and freezing temperatures. Hence, it can be used to make trays and packaging for frozen dinners.
Renewable Energy: PET films are used as substrate material for thin-film solar cells.
Polyester Prototypes using CNC, 3D printer machines, and injection molding
CNC machines
Polyester is a great material for use with CNC machines. It comes in sheet stock and works well with a machining process that involves a mill or lathe. It is transparent and shatter-proof, all of which translates into great machinability. Polyester used with CNC machines is normally transparent, but it can be colored as well. Sometimes, additional processing of the polyester is required after it has been used with a CNC machine in order to remove any tool marks and scuffs.
3D Printing
Polyester is also available in a filament form which can be used with 3D printers to create prototypes using computer-aided design files. As polyester is a thermoplastic which can be melted and then cooled without degradation, it is used with 3D printers. It is a great material for 3D printing because polyester is flexible and tough. Polyester also gives out less odor while 3D printing that ABS and PLA.
Injection Molding
Polyester is available in pellet form which is ideal for use with an injection molding machine. As polyester is a thermoplastic, the pellets can be melted and liquefied. The liquid polyester can then be injected in a mold with pressure. One precaution that needs to be taken with polyester is that it must be dry before use since the material is hygroscopic. Shrinkage for PET or polyester is very low, which is another desirable quality.
Disadvantages of Polyester
- Polyester might be recyclable, but it is not bio-degradable. Hence, there is always the issue of accumulation of vast quantities of polyester and PET.
- PET is at times susceptible to the process of oxidation. If a food item such as wine or beer is stored in a PET container/bottle, then the oxidation might actually degrade the taste of the beverage. Storing any food/drink item long term is not recommended with PET containers.
- Polyesters can be affected by hydrolytic degradation. Hence, they must be dried using a dehumidifier before processing.
- Polyester is not a carbon neutral material. It is made from hydrocarbons and hence it fossil fuel dependent. It is also affected by the price of oil.
Properties and Specs
| Property Type | Detail |
| Scientific Name | Polyethylene Terephthalate |
| Resin Identification Code | 1 |
| Chemical Formula | (C10H8O4)n |
| Dielectric Constant | 3.0 |
| Specific Gravity | 1.56 |
| Melting Temperature | 260oC |
| Flexural Strength | 221 MPa (32000 PSI) |
| Maximum Continuous Use Temperature | 132oC |
| Shrink Rate | 0.1 – 0.3% (0.001 – 0.003 in/in) |
| Izod Impact Strength | 13 – 35 J/m |
| Elongation at break | 13% |
| Rockwell Hardness | M94 – M101 |
| Poisson’s Ratio (v) | 0.37 |
| Heat Deflection Temperature | 70oC at 66 PSI |
| Injection Mold Temperature (Typical) | 74oC to 91oC |
| Chemical | Resistance Level |
| Acid (Concentrated) | Good |
| Acid (Dilute) | Good |
| Alcohol | Good |
| Alkalis | Poor |
| Aromatic Hydrocarbons | Poor |
| Greases and Oils | Good |
| Halogenated Hydrocarbons | Poor |
| Halogens | Good |
| Ketones | Good |


Tensile strength of Polyester is shown as 22,000 psi.
Other sites seem to show about 1/10th eg c 2300 – 4500
(?)
T Straw